AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Hollow cathode lamp block diagram1/17/2024  MonochromatorĪ monochromator is an optical device that transmits a narrow band of wavelengths of light or other radiation from a wider range of wavelengths. These types of lamps are widely used in atomic absorption spectroscopy instruments. Such types of lamps facilitate for determination of samples without change of lamps each time. Such types of lamps are called multi-element hollow cathode Lamps. Several types of hollow cathode lamps are available in our market. The lamp forms a glow of emission from the hollow cathode. The lamp is filled with noble gas at low pressure. The metal which is used in the cathode is the same as that metal that we analyzed. Therefore, we used a chopping wheel between the radiation or pulsed potential. Radiation from the hollow cathode lamp should not be continuous due to spurious radiations. The hollow cathode lamp has two electrodes, one is cup-shaped and made of a specific element. We used a hollow cathode glow discharge lamp to give sharp emission lines for a specific element in atomic absorption spectroscopy instrumentation. The extreme narrowness of the absorption line in the sources causes problems. We need a continuous source of radiation in the AAS instrument. The sample is aspirated through the air into the mixing chamber. A ribbon flame is produced in the AAS instrument. At too high temperatures, atoms can be ionized.įuel and oxidant gases are fed into a mixing chamber which passes through baffles to the burner. The temperature should be controlled very carefully for the conversion of atomic vapor. 
Heat energy is utilized in atomic absorption spectroscopy to convert metallic elements to atomic dissociated vapor. Any atomic absorption spectroscopy instrumentation has the following types of components,Ītomization can be carried out either by a flame or furnace. Atomic absorption spectroscopy instrumentationįor instrumentation, flame, non-flame, and graphite furnace are available in atomic absorption instruments. Such high temperatures can be attained by using an oxidant in the flame along with fuel gas in atomic absorption spectroscopy.įor example, we used oxyacetylene flame for the analysis of aluminum, titanium, and rare earth elements in an AAS instrument. Therefore, we need a high temperature for the vaporization of such metals. Some metals are not easily atomized or vaporized. These metals are easily vaporized at low temperatures. We used low temperatures for metals like copper (Cu), lead (Pb), zinc (Zn), and cadmium (Cd). Usually, natural gas, propane, butane, hydrogen, and acetylene are used as fuels to create a flame.Īir, oxygen, nitrous oxide, and a mixture of nitrous oxide and acetylene are used as an oxidant for flame creation in atomic absorption spectroscopy.Ī list of various flames with maximum temperate is given below the table, Atomic absorption spectroscopy flame We used fuel and oxidant to create an atomic absorption spectroscopy flame. 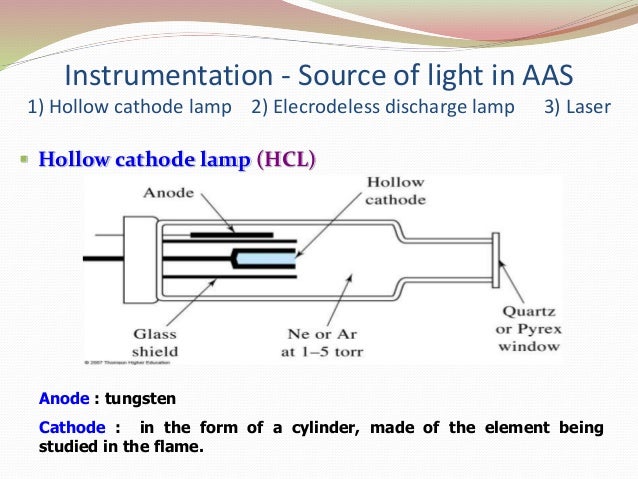
Usually, lines arising from the ground state are almost important in atomic absorption spectroscopy. From the ground state of an atom is excited to a higher energy level by absorption of energy.Ītomic spectra are identified by sharp lines which can be distinguished from broadband spectra associated with molecules. The electronic transition is specific to a particular element. Light at this wavelength has absorbed energy to excite another electronic state. Elements with low excitation energy can be determined by flame emission while high excitation energy can be determined by atomic absorption spectroscopy.Ītoms absorb light at a definite wavelength depending on the nature of chemical elements. Many difficult and time-consuming instruments were replaced after the discovery of AAS.Ītomization, hollow cathode lamp, monochromator, detector, and recorder are the main components in atomic absorption spectroscopy instrumentation. In 1955 Alan Walsh from Australia applied the principle and instrumentation of atomic absorption spectroscopy for the analysis of chemical elements. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
